Importers of consumer electronics need to understand the types of batteries for electronic products that are on offer so that you can select the correct type for your needs and consumers.
Before we explore the different types of batteries, let’s look at the market for consumer batteries which is set to reach $50 billion by 2025. As devices continue to play a ubiquitous role in consumer’s lives and new types of electronic products become popular (such as IoT devices, wearables, etc), battery knowledge is increasingly important for the creators and importers of electronics.
The overall global battery market including commercial and industrial applications already exceeds $100 billion and is set to grow by around 14% per year between 2020 to 2025, with the need for LI-ion batteries by EV auto makers, in particular, driving this growth.
Since a lot of our clients have had to educate themselves about the different types of batteries, we’ve created this guide to outline the basics.

TL;DR
This guide breaks down the most common types of batteries used in electronic products, including primary (non-rechargeable) and secondary (rechargeable) chemistries, and explains how they compare on energy density, cost, cycle life, safety, and typical use cases. It covers options like alkaline, lithium-ion, NiMH, lead-acid, coin cells, and speciality chemistries, and helps product developers choose the right battery based on performance needs, form factor, runtime, and regulatory or safety requirements, so you can balance cost, reliability, and user experience.
Table of Contents
Please keep reading, or select the section you want to skip to by hitting the links below.
How many different types of batteries are there?
So, batteries are only becoming more commonplace every day and with this in mind, let’s explore the different types of batteries, their features, benefits, costs, and more.
Batteries can be organized into two main categories:
Within the primary and secondary battery categories, they can also be roughly organized by shape, commonly taking on these formats:
Cylindrical cells – this is the ‘traditional’ battery shape most think of and exists in both primary and secondary forms.

Button/coin cells – often primary batteries used in small portable devices such as watches.

Prismatic cells – these are usually the slim, rectangular-shaped secondary Li-ion batteries commonly used in laptop computers.

Pouch cells – an example is Li-po secondary batteries used in cell phones.

If we consider electronic products, it’s feasible that you will provide products with different types of batteries of either type, or perhaps on a ‘batteries-not-included’ basis, too.
Each battery type has multiple power capacities, and in the following sections, we’ll go into that.
Primary Batteries
Which primary batteries are commonly used in electronic devices today?
Today’s ‘disposable’ primary batteries that consumers are likely to find on the average supermarket shelf are commonly two types:
Alkaline batteries

Alkaline batteries are made from zinc metal and manganese dioxide with an alkaline electrolyte of potassium hydroxide. These are favored over other primary types (such as zinc-carbon) as they have a longer shelf life of around 2 years and higher energy density providing an output of, averagely, around 1.5V.
To give some idea, a common AA alkaline battery has a power capacity (or size) of between 1,800 to 2700mAh. This isn’t far away from a modern smartphone battery’s capacity, although it’s single-use.
Big-name brands such as ‘Duracell’ and ‘Panasonic’ generally provide alkaline batteries in-store and ‘premium’ versions are available which have a higher voltage.
They come in the following sizes:
Benefits
- Low-cost
- Easily obtainable
- Available in a variety of sizes for different applications
- Long shelf life of 5-10 years
- Discharge over time is minimal due to low internal resistance – only 0.17% per month
- High power capacity for a primary battery
Drawbacks
- Single-use
- Not particularly environmentally-friendly as most call for being thrown in the trash after use
- Do not provide long-term power and are a poor option for power-hungry devices
- Can be bulky
- Replacement cost mounts up over time in comparison with secondary batteries
Lithium batteries

Lithium batteries benefit from a far longer lifespan of around 10 years which is why they’re often used in watches and medical devices where changing batteries regularly isn’t convenient.
Most consumers are familiar with them as single-cell button batteries, but they can also be found in the regular battery shapes (such as AA, AAA, etc).
The average lithium battery will provide a power output of anywhere between 1.5V (similar to alkaline batteries) to 6V+ depending on the size. Just know that, in general, lithium batteries are a far greater source of energy than their alkaline cousins. This is reflected in the cost, though, so most cheaper electronics will not use them as overall costs will become prohibitive.
For example, one lithium AA battery can cost around $10, whereas you can purchase a 50 pack of regular zinc alkaline batteries for around the same cost!
Lithium batteries are not to be confused with Lithium-ion or Lithium polymer batteries. The latter two are secondary rechargeable batteries.
Benefits
- Easily obtainable
- Available in a variety of sizes for different applications
- Often very small and so used in items like watches and hearing aids
- Long-lasting in comparison to other primary batteries
- Small cells can be stacked to create high voltages
- Long shelf life
- Discharge over time is minimal due to low internal resistance
- Small and lightweight
Drawbacks
- Single-use
- Not particularly environmentally-friendly as most call for being thrown in the trash after use
- Can be quite expensive
- May require a special mount
Zinc carbon batteries

Zinc carbon primary batteries are old technology and provide far less power capacity than alkaline batteries with the average AA type holding only between 400 to 1,700mAh.
As with other alkaline batteries, they’re cheap, easy to purchase, and have a low self-discharge rate of only around 0.32% per month.
Their shelf-life is not as impressive as alkaline batteries, as over time they’re prone to oxidation and leakage and so will only last for around 2 years.
This isn’t all bad, as they’re tried-and-tested and are suitable for use in smaller electronic products which have a low power draw, such as a wall clock.
Since they have largely been superseded by alkaline batteries, they aren’t as readily available in-store as they used to be, nonetheless, consumers can still purchase them cheaply enough, with a 20 pack of AA zinc carbon batteries costing around $4.50.
Benefits
- Easily obtainable
- Available in a variety of sizes for different applications
- Discharge over time is minimal due to low internal resistance
- Low cost
Drawbacks
- Single-use
- Not particularly environmentally-friendly as most call for being thrown in the trash after use
- Poor overall power capacity
- Short shelf life compared with other primary batteries
Which types of products commonly use primary cells?

Cheaper electronics and those which do not require a lot of energy to operate often benefit from using primary cells, as the consumer will have no problem with occasionally purchasing disposable batteries.
In fact, including rechargeable batteries could increase the price of, say, a small digital clock with a low power draw to the point where its cost is no longer competitive in the market, so using secondary cells is simply not feasible.
Commonly seen in toys, Christmas lights, torches, TV remote controls, and more.
What are the advantages of primary batteries?
Primary batteries do not get the attention of secondary rechargeable batteries, as the ability to reuse the latter makes them more media-friendly and attractive to consumers who want a greener option.
However, primary batteries still have an important role to play in the electronics industry.
They will be advantageous in the following applications:
Batteries
- When recharging is inconvenient of impossible
- If the device in question has a very low power draw and will be in operation for a long period of time
- When the batteries need to be stored for a long time before use, but still need to be reliable and provide high energy when called upon
- If there is a need to transport batteries easily
Secondary Batteries
Which secondary batteries are used in electronic products today?
Secondary batteries found in electronic products are rechargeable and may not always be readily purchased by consumers (in the case of internal cells for laptops and other in-built batteries). These different types of batteries are commonly seen in this market:
They usually include a battery management system to assure that they operate within safe and normal parameters in order to protect the battery, retain optimal lifespan, and avoid safety issues such as fires (which could potentially happen with secondary batteries like Li-ion if they are abused).
Let’s start with the two types of secondary batteries that are often integrated into a product (such as cell phone batteries).
Li-ion batteries

Li-ion batteries are the most common high-capacity secondary batteries used in today’s power-hungry devices such as laptop computers, mobile phones, cameras, and more.
Li-ion battery technology is popular for use in today’s electronic devices as it provides high power density, doesn’t self-discharge quickly, and is relatively affordable.
They are typically made from 3 pieces:
- Positive electrode (cathode) made from a type of lithium oxide
- Negative electrode (anode) made from carbon
- Solvent electrolyte with lithium salt
Because a worst-case scenario is for lithium-ion batteries to combust in certain circumstances, most Li-ion batteries used in everyday electronic products will also feature an electronic controller to regulate the energy flow to prevent the battery from overheating or exploding.
To prevent Li-ion safety issues avoid bending them, exposing them to high temperatures, wrongly connecting, and overcharging them, as all of these circumstances could lead to combustion.
While the types of lithium ion chemistries used provide varying battery performances, in general, Li-ion batteries suffer from an average lifespan, poor thermal stability, and sensitivity to overcharging which can damage the battery and cause fires.
Types of Li-ion chemistry
- NMC (Lithium nickel manganese cobalt oxide) – the most commonly used Li-ion battery type. Used in all kinds of portable electronics and power storage applications.
- NCA (Lithium nickel cobalt aluminium oxide) – famously used in Tesla EVs, these batteries are an alternative to NMCs and can also be found in numerous consumer electronics, particularly those from Japanese manufacturers Panasonic and Sony.
- LCO (Lithium cobalt oxide) & LFP (lithium iron phosphate) – older technologies, but still seen regularly. Superseded for the most part by NMC batteries until recently when LFP is making a comeback in larger cells, particularly those used in EVs.
Benefits
- High power density (they range anywhere between around 3,500mAh in a phone, up to around 20,000 mAh in laptop batteries and power banks)
- They always remain easy to charge
- They are mainly unaffected by a reduced total amount of charge over time
- Low self-discharge of only 1.5-2% per month when not in use
- Quite high cell voltage of 3.6V during operation
- Cutting edge LFP prismatic batteries used in EVs can hold 200+ Ah
Drawbacks
- They will naturally degrade and start to hold less charge over time
- Battery lifespan is often just ‘OK’ in comparison to other technologies
- Flammable in extreme circumstances, such as when damaged
- Battery management system required for safe operation
- Costs are kept high by the scarcity of cobalt and nickel
- Not very durable
- They perform poorly at very low temperatures (below 0°C)
Li-po batteries

Lithium-ion polymer batteries are at the cutting edge of secondary battery technology used in electronic products. Rather than using a liquid electrolyte, a form of highly conductive polymer in dry, gel, or porous form is used which allows greater energy storage and smaller battery size.
Because they allow batteries to be made very thin, light, and to conform to different shapes, but still have a high output, they’re used in premium laptops, drones, tablet PCs, and other higher-end electronics.
The difference between Li-po batteries and other Li-ion types is that rather than using a liquid electrolyte, there is a polymer (plastic) component to theirs which, in many cases, is like a gel. Using a more solid electrolyte in a pouch cell makes the battery stronger, less likely to leak, and, therefore, safer.
NASA, EV companies, and various other manufacturers are even developing fully ‘solid state’ Li-po batteries using solid polymer or ceramic electrolytes.
A typical LiPo cell will produce a nominal 3.7 volts of power. Multiply the 3.7 volts per cell, in the case of a multi-cell battery. In terms of overall capacity, they vary wildly from anywhere around 300mAh up to 20,000+mAh (similar to regular Li-ion) although in general Li-ion batteries will offer a larger capacity for lower cost.
These batteries can be purchased from specialist stores, but most consumers will purchase products where they are already integrated into the product, such as a mobile phone or laptop (the new Apple MacBook Pros use them due to their low profile thickness). Therefore it’s up to the manufacturer to select and use the type of Li-po battery that they desire for the product and assess whether the cost is worthwhile.
Benefits
- Far thinner than regular Li-ion batteries
- Shape & size can be customized to conform to specific dimensions
- Lower chances of leakage and fire
- Good power capacity, a little less than that of Li-ion
- Low self-discharge of only 1.5-2% per month when not in use
- Quite high cell voltage of 3.6V during operation
Drawbacks
- Shorter lifespan than regular Li-ion batteries
- Capable of holding less power than other Li-ion batteries
- Far more costly than Li-ion batteries
- Can be flammable if not used properly
- Will lose capacity gradually over time
LiFePO4 batteries
LiFePO4 (LFP) is another type of Lithium-ion technology which uses Lithium phosphate cathode cells, but are otherwise fairly similar to the other Li-ion batteries in terms of performance as they also provide good energy capacity.
There are some key differences, though.
They don’t provide such a high cell voltage, have greater self-discharge, and have a reduced power capacity in comparison to Li-ion and LiPo batteries, hence why the latter two are more popular for use in today’s electrical products.
But they’re a safer battery overall as they are less affected by overheating and damage and the phosphate is not flammable, while also providing a longer lifespan in general than the others. This is due to their different cathode material where a non-flammable phosphate is used.
Due to their long lifespan, series of 4 (12.8V) or 8 (25.6V) cells are now used as a Lead-acid battery replacement in vehicles as they’re lighter, last longer, and have a higher capacity.
Benefits
- Lower cost than regular Li-ion batteries
- Safer than Li-ion batteries without the same overheating and fire risk
- Lower loss of capacity than Li-ion
- Very long lifespan of around 10 years (2-3k more charging cycles than Li-ion)
- Constant discharge voltage allows battery to deliver full power at all times
Drawbacks
- Capable of holding less power than other Li-ion batteries
- Lower cell voltage of 3.2V
- Higher self-discharge rate of 4.5% per month compared with Li-ion and Li-Po when unused
The next two secondary battery types are more likely to be those purchased separately from products by consumers from stores. These are not as efficient as Li-ion or Li-po batteries, but they are less costly.
NiMh batteries

NiMh batteries have effectively replaced Ni-Cd batteries for most of the same applications as they’re newer technology, usually have a far higher energy capacity, and are less environmentally harmful.
Instead of the toxic Nickel and Cadmium combination, they use nickel metal hydride and are also often cylindrical and can be found in most standard sizes (such as AA, AAA, etc). Due to being less toxic, the EU’s 2006 Battery Directive encourages their use in portable electronics.
Although Li-ion batteries are still preferable today, for smaller portables which require standard battery sizes (let’s say a digital camera, for example), NiMh batteries are a better alternative long term than disposable primary batteries.
One drawback with standard NiMh batteries is that they suffer from quite a high amount of self-discharge meaning that an unused device using these batteries as a power source that has sat for a while may need to be charged again before use which consumers will find inconvenient. This issue has been solved in the newer low self-discharge nickel metal hydride batteries (LSD NiMH). Whereas standard NiMh batteries can lose up to 30% of their charge per month when not used, LSD NiMh batteries can retain charge approximately 6 times longer. They must also be charged carefully, as overcharging can damage them and affect their capacity and performance.
Pricing is similar to that of NiCd off the shelf, although consumers will probably find NiMh batteries more readily – for instance, if you search, say, Amazon most ‘rechargeable batteries’ are NiMh and there are fewer NiCd options.
They have a larger capacity to NiCd, with AA holding between 800 to 2,700mAh of energy.
One benefit of note is that they are cheaper and tend to hold their charge fairly well in comparison to their NiMH cousins that have mainly replaced them.
Benefits
- The average NiMH battery has about 2-3 times the capacity of NiCa/NiCd.
- Better for electronics with a higher power need than NiCd or disposable batteries
- They are more efficient than alkaline batteries
- The LSD NiMH has a very low self discharge compared with NiCd and Li-ion
- Greener option as they can be recycled and do not include toxic cadmium
- Easy to obtain online and in-store
Drawbacks
- The normal NiMH batteries have a
- High-Self-Discharge. They lose about
- 30% of their charge every month which means they’re not good for use in devices which require a low power draw over a long period of time, such as a smoke alarm
- Higher initial purchase cost to alkaline batteries may be off-putting for consumers
- Suffer from memory loss over time resulting in a reduced capacity, although less than NiCd
- They aren’t as durable as other batteries – sensitive to overcharging and drop shock
Ni-Cd batteries

These batteries are made of Nickel and Cadmium combination and are often cylindrical and can be found in most standard sizes (such as AA, AAA, etc).
They aren’t widely used these days as they’ve been largely replaced by more efficient NiMH and Li-ion batteries, although can still be found and purchased for use in portable electronics.
Their capacity is generally quite a lot lower than NiMh, with an AA version holding only between 600 to 1,000mAh. Pricing will be from somewhere around $20 for four AA Ni-Cd batteries up to $70 for a larger battery pack, although you will mainly see them in specialist online stores rather than being able to purchase them off-the-shelf.
One benefit of note is that they tend to hold their charge fairly well in comparison to their NiMH cousins.
Benefits
- Low cost for rechargeable batteries
- Fairly low discharge rate of around 10% per month when not being used
- Readily available in standard sizes which makes them a good alternative to single-use alkaline batteries
- One of the hardest secondary batteries to damage through incorrect charging
Drawbacks
- Old technology compared to more modern NiMH and Li-ion batteries
- Poor performance in power-hungry portable electronics (Portable MP3 players, cameras, gaming systems, etc)
- More expensive initial cost to alkaline batteries
- Prone to memory loss and take on less charge over time
- Not green as the chemicals used are highly toxic and are difficult to dispose of
- Poorer capacity compared with NiMh
Which types of products commonly use secondary cells?

Any electronics with a greater power demand, like tablet PCs with a 2A power draw when charging, won’t work well with a primary cell. Also, one must consider the design of the product. Having an aperture and space for replacing disposable batteries necessitates a large ‘battery pack’ to be included within, or connected to, the device which could add a lot of bulk. Anyone who remembers the old days when battery-powered stereo systems with CD players first came out will know that primary cells don’t last long in larger electronic devices!
What do you need to know about battery capacity?
What does battery capacity in Milliampere-hours (mAH) mean in relation to consumer electronics?
Battery capacity refers to the amount of energy stored in the battery (the size of its ‘gas tank’). We use mAH, which is an abbreviation for Milliampere hour, to measure the amount of energy capacity or how long they can operate for.
Most of today’s consumers have been conditioned to understand the mAH figures given for ‘smaller’ batteries such as those found in mobile phones and other portable electronic devices.
For instance, an Apple iPhone 11 has a Li-Ion 3110 mAH internal rechargeable (secondary) battery. So let’s say, for example, if the iPhone requires 100 mA to run, this means that the battery will last around 31 hours. (3100mAH / 100 mA = 31 hours)
Of course, the battery is running the screen, antenna, processor, camera, etc, at any given time and so the battery could last significantly less than this from full to empty, so the consumer knows that the higher the mAH figure, the better.
In terms of size, a single AA battery may hold 2000 mAH of energy, whereas a prismatic Li-ion cell may hold 10,000mAH. The latter is clearly larger and more expensive in turn, as you’re essentially paying for a larger ‘gas tank.’
Where can we see the battery capacity?

Most manufacturers go with battery life in hours based on average usage and that’s helpful for the consumer.
Primary batteries often don’t bother with an mAh figure, rather suggesting an average lifetime in hours.
However, for most electronics using secondary batteries, for instance, a laptop or cellphone with a Li-ion battery, it’s easy enough to find this figure (for instance, here’s technical data for the iPhone 11).
There is no great need to dig around for battery capacity data, though. Wikipedia has a great page devoted to it for the different types of both primary and secondary batteries of all kinds here which you should bookmark for reference.
When YOU need to know about battery size and capacity

If you’re bringing electronics to market measuring the power requirements of the device will occur during the R&D stage. It will become clear what level of power capacity your battery is going to need in order to give the device an acceptable, or maybe great, operating lifespan between battery changes or charges.
If the device is made to run on primary batteries, it’s simply a case of assessing what kind of battery will be most suitable in terms of power capacity and size, for example AA or AAA.
However, if you’re producing a device that requires a rechargeable (secondary) battery, you’ll need to balance capacity, cost, and size with your consumer’s reasonable expectations for a decent operating time.
FAQ
Battery sizes are usually denoted by a combination of letters or numbers. As an importer, you will need to decide which size is most appropriate for your product.
Secondary cells in today’s electronic devices are often prismatic or pouch cells which can be bespoke sizes and, therefore, don’t appear in this list of standard battery sizes (although some rechargeable batteries on the market, such as NiCd AAs, for example, will adhere to these standard sizes):
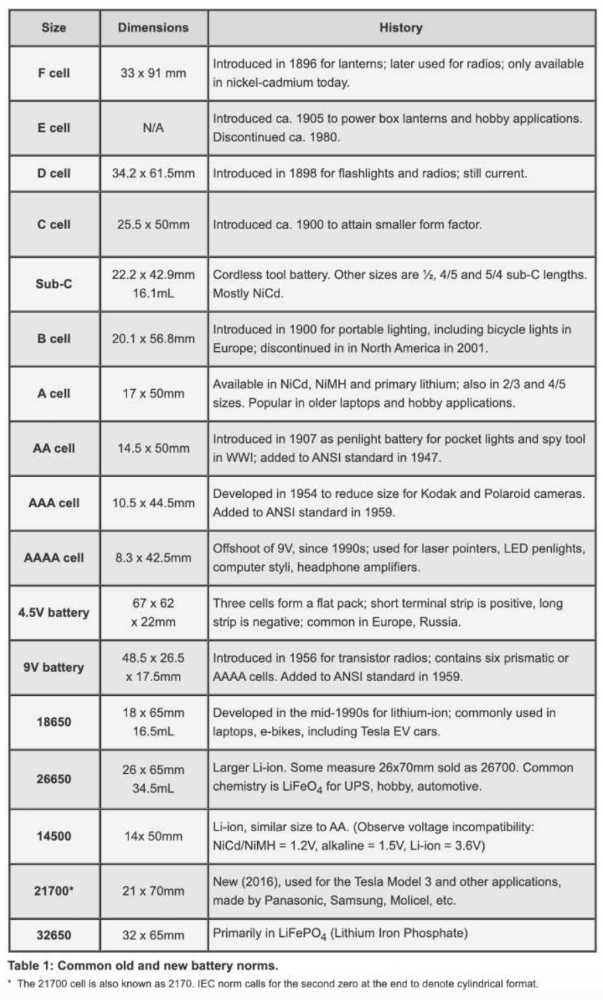 Table courtesy of Battery University.
Table courtesy of Battery University.
For American importers, we’ve created this whitepaper exploring the Lithium Ion Battery Safety Guidelines For US Imports. Just hit the button below to read it – no download required!
The testing that you do on batteries is dependent on the level of risk you’re willing to tolerate and any mandatory requirements for your market.
Let’s assume you want to assure Li-ion battery quality, reliability, and safety coming out of China with your products. Here are some notes on what we’d suggest as a rough example:
- How can the Li-ion batteries be tested in order to assess their quality/capacity and limit the risk of poor performance during use (aside from user actions like over-frequent charging, etc)?
We suggest performing the following:
– Test life time in the product (force discharge with max usage)
– Test battery performance in different temperatures
– Measure battery self-discharge - Does Sofeast have any standard tests for batteries?
Yes, aside from the 3 above, we also test to the UN38.3 standard for safe shipping. - What’s the best way to be sure about battery quality/capacity, etc?
UN38.3 should give a good understanding about the battery quality alone, but we would also recommend cycling and temperature testing.
You should visit this page where we collect information about various battery tests, or watch the videos below:
Charge-Discharge Cycle Tests On Batteries For Electronic Products https://youtu.be/ZvZQxWqRb54
Battery Capacity Tests For Importers Of Chinese Electronics
https://youtu.be/h74MSLIdxuA
Battery Thermal Discharge Tests For Electronics Importers
https://youtu.be/aAlyJp463sM
Battery Life Cycle Tests For Importers Of Electronic Products
https://youtu.be/NXsGDiXyQhI
When producing battery-powered products as the importer you are mainly responsible for the safety and compliance of the batteries (and products in general) coming into your market (it’s also possible that the manufacturer may share responsibility if this is included as a clause in your manufacturing agreement.
Assuring that your batteries comply with the necessary safety standards is a step in the right direction, and in order to do so, you need to know which standards apply to your market.
General battery standards
Here are a number of the standards that cover both primary and secondary batteries. If you aren’t including primary batteries as a part of your product or are a battery manufacturer, knowing the standards regarding primary cells are less crucial.
- The EU Battery Directive 2006/66/EC regulates the batteries coming into the EU market and outlines the restrictions on the use of toxic heavy metals, recycling, and labeling.
- RoHs 3 this ‘Restriction of Hazardous Substances’ directive controls and limits hazardous substances which can be used in batteries, cadmium in particular, and is generally used globally.
- Waste Electrical and Electronic Equipment recycling (WEEE) mandates the treatment, recovery and recycling of electric and electronic equipment and is required for the EU market.
Lithium battery standards
Because Lithium batteries have specific safety concerns, especially secondary Lithium-ion types, let’s pay special attention to some of the key standards to test to here.
- IEC 62133-2:2017 – Safety requirements for portable sealed secondary lithium cells, and for batteries made from them, for use in portable applications – Part 2: Lithium systems – this is a fairly common standard for lithium-ion battery safety including overcharging, case stress, short circuit, and drop testing.
- UN/DOT 38.3 6th Edition, Amendment 1 – Recommendations on the Transport of Dangerous Goods – this safety standard must be met if Lithium metal or Lithium ion batteries are to be shipped by air, sea, or rail.
- EN 60086-4 – Safety of primary lithium batteries – a European standard that describes tests and requirements for primary lithium batteries to ensure their safe operation under intended use and reasonably foreseeable misuse.
- EN 62133/UL62133 – Safety requirements for portable sealed secondary cells, such as lithium-ion – a European standard concerned with the tests and requirements for rechargeable lithium cells, such as Li-ion and Li-po batteries.
- ANSI C18.2M, Part 1-2019 American National Standard for Portable Rechargeable Cells and Batteries—General and Specifications
- IEEE 1625 – Standard for Rechargeable Batteries for Multi-Cell Computing
- IEEE 1725 – Standard for Rechargeable Batteries for Mobile Telephones
UL standards call for strict testing for battery-powered products and are mandated for the US market. These standards, relevant to Lithium-based batteries in particular, might be useful. - UL 1642 – Standard for Safety for Lithium Batteries
- UL 2054 2nd Edition – Household and Commercial Batteries
- UL 2056 – Outline of Investigation for Safety of Power Banks
- UL 2595 – Standard for Safety for General Requirements for Battery-Powered Appliances
- UL 4200A – Standard for Safety for Products that Incorporate Button or Coin Cell Batteries Using Lithium Technologies To learn more about the US standards, read about the CPSC Battery Standards which is an explanation about the standards for the US market (that may also be relevant elsewhere).
- UL 8139 – Electrical Systems for Electronic Cigarettes – with the proliferation of e-cigarettes and vapes, this standard is commonly used and worth being aware of due to the safety concerns about these products which can cause injury easily if problems occur with the batteries.
Firstly, as the importer, it’s highly unlikely your liability for any safety issues occurring due to batteries in your products would be nil.
However, testing the batteries at the battery supplier’s factory (this qualifies them before production even starts) and then during production on samples from the right batch, if their standard is clear and their results are documented, it would definitely help you in case you are sued for a problem the test was aiming at detecting. You will probably have covered yourself against the accusation of negligence which would hurt you in a court of law if proven correct.
Note: It’s important for the samples to be selected randomly from production by your representative (rather than picked by the manufacturer). That’s a plus since this usually prevents a dishonest manufacturer from gaming the system by, say, swapping out the batteries you’re expecting for cheaper versions in order to increase their profit margin.
As ever, in this situation, it’s best to consult with a lawyer to get guidance on your liabilities
Yes, it is possible.
Since batteries are quite expensive either as a standalone item or component, there is always a temptation for some suppliers to switch them out for cheaper equivalents or even use cheaper fakes.
For obvious reasons, this is a compliance and safety issue, as a battery that is not as expected will similarly not adhere to the standards expected, certainly not in the same ways anyway.
What can be done to reduce the risks of this happening?
There are a number of actions to take if you feel you’re at risk of a supplier playing this kind of game:
- Check the battery specification sheet they provide against the results of testing the suspect batteries
- Contact the battery manufacturer (for example, this might be Panasonic or Samsung) and ask them to confirm if the battery codes and information provided are genuine
- Make sure that it’s clear in your quality plan what your expectations are for the battery, so there can be no ambiguity (this helps show a supplier’s wrongdoing if they have tried something unacceptable)
- Add to your manufacturing agreement that a specific battery must be used and that alternatives of any kind are not acceptable
Beyond testing the batteries you receive for quality, safety, and authenticity, having legal recourse to take action if a supplier doesn’t provide the promised batteries is a good incentive for them to abide by the agreement.
Batteries for electronic products store DC power as AC cannot be stored.
The best way to think about AC and DC currents are in terms of distance. If you want to transport power over long distances, such as from a power station along the grid into homes, the higher-voltage AC will be used. Whereas for short distances, such as from an electronic device’s internal battery to powering the device, DC is preferable as it can be stored and used when required.
Larger electronics such as a laptop computer will be run on both battery and mains power, and in these cases the device will come with an AC-adapter of some sort which will take the AC current and convert it into DC which the devices run on.
Watch this video for a scientific explanation of the difference between AC and DC currents:
Read more:
Rechargeable batteries found in electronic devices, autos, and anywhere else, usually require a battery management system, or BMS, which is a small circuit that is incorporated into the battery.
Given the sensitivity of some secondary batteries, like Li-ion, to incorrect charging or discharging practices, the need for a BMS becomes clear as they can be critical when it comes to safety.
A BMS is chiefly used to monitor and control the criteria which affect battery life and safe function. Every rechargeable battery has conditions that must be maintained in order to operate safely, such as a certain voltage, current, and remaining at a safe temperature.
Operating outside of these conditions could lead to problems such as over or under voltage that may negatively affect the batteries lifespan, cause the battery to stop working correctly, or, in extreme cases, cause dangers such as overheating and fires:
What does the BMS measure?
This can vary, but the BMS and its software will be measuring battery performance in order to avoid it entering dangerous operating conditions.
They may measure:
- Battery State Of Charge (SOC) – controls safe charging and discharging of the battery to avoid overcharging or discharging, the latter being very harmful to Li-ion batteries.
- Battery temperature – some BMS will monitor battery temperature and if it reaches a certain prohibited level can activate a thermal management system in order to cool it.
- Battery State Of Health (SOH) – charge/discharge cycling can help calculate this to show wear and tear in the battery and reduced total capacity.
- Current coming into or flowing from the battery.
- Battery State Of Safety (SOS)
If the battery is found to be operating outside of normal conditions, what can the BMS do?
The BMS can activate cooling measures, switch off the battery, or reduce or stop the device’s power use.
There are 2 documents that you need to obtain from a battery supplier wherever possible:
If your battery supplier cannot or will not provide you with this information, that’s a major red flag. You should consider these to be ‘the basics’ when it comes to purchasing batteries for use in today’s products.
Knowing how the batteries are made, their ingredients, and how they should be stored, handled, and shipped is important information for your own compliance.
Furthermore, if you do go on and get 3rd party lab tests done on the batteries, providing the testing lab with these will also be helpful for them and you can cross-check against what the supplier has provide in the spec sheet to assure that they’re providing the right type of battery with the correct voltage, etc, as promised.
Batteries may also be categorized by the number of volts that they provide. For instance, a 9-volt battery provides 9 volts of energy when in a circuit.
To know what voltage your battery is, check the battery specification sheet. This will tell you important information including voltage when fully charged and help you to know if the battery in question is right for your needs.
As an example, primary ‘AA’ batteries usually have a voltage range of between 1.2-1.5V. As a battery is used its voltage decreases, and you’ll see that battery-powered devices using primary cells will start to slow down and stop as the battery approaches the end of its life.
But this is not the case with all types of batteries. Li-ion secondary cells provide a constant discharge voltage rate, meaning that they will operate at full power until totally empty. This is why they’re used in devices like cell phones and laptops which require relatively high and stable power to operate in comparison to a simpler device such as an analog desk clock which will be fine with primary cells which eventually slow down and die.
As a buyer of batteries, you need to know the voltage required for your product’s circuit. After you know this, you will then know the battery voltage needed.
Today, the most commonly used batteries for portable electronics are Li-po lithium polymer batteries, usually in a rectangular shape.
Despite being more costly than prismatic cells, their thin size and light weight make them preferable for today’s portable electronics where thickness and weight are ever-decreasing.
After you have decided which batteries you need an essential quality test on incoming batteries from your supplier is to check their voltage. Then you’ll quickly and easily know that they hold the required amount of power.
Batteries are meant to store a certain amount of voltage and this can be tested by connecting it to a multimeter (or voltmeter).
You’ll set up the multimeter and select the DC voltage setting (a V with 3 dots or a dash above it or DCV or something similar).
Most digital multimeters used today will automatically recognize the battery’s voltage range, so all you need to do is hold the positive and negative probes to the correct terminals – in this case, positive is red and negative is black.
The battery’s voltage will appear on screen and should, if the battery is full or almost full, be around the correct voltage. So, for a 9 volt battery, you should be seeing a value of 9 volts or just under.
If the batteries you receive from a supplier do not reach the required voltage, they fail the test and you can take action to solve the issue.
Other tests will be required to assess battery-safety, such as for Li-ion batteries which are due to be shipped.
Watch a video explaining how to test battery voltage using a watch button cell as an example here:
These two most popular secondary batteries may seem hard to separate when choosing a suitable battery for electronic devices that require a lot of energy in a rechargeable package. But there are some key differences to consider.
Li-ion batteries
Lithium-ion have become the benchmark in rechargeable batteries for electronics which require a lot of power due to their high power density, resistance to ‘the memory effect’ (when batteries progressively take on less charge as time goes by), and their low purchase cost. They also retain charge well when unused, having a fairly low self-discharge rate.
However, they have been dogged by safety issues which were brought into the public’s consciousness with the Samsung Galaxy Note 7 recall due to the phones’ Li-ion batteries catching fire. Li-ion batteries can be prone to catching fire if damaged, incorrectly charged, or stored in the wrong conditions. They are designed in such a way as the positive and negative electrodes cannot come into contact with each other, but if damaged that can change and cause fire.
They also age and slowly retain less overall charge over time, although this is not enough to prevent them from being an effective power source for a number of years for most devices.
Li-po batteries
Lithium-polymer batteries are the ‘newer kid on the block’ and are much loved by manufacturers of premium electronics or devices where space is at a premium as they are thinner and lighter than Li-ion batteries while providing a similar (slightly lower) power capacity.
They do not suffer from the same fragility that Li-ion batteries do, as they don’t use a liquid electrolyte which could leak upon damage. Because they don’t require a rigid shell for protection they’re around 20% lighter and can be formed thinner, too.
What makes them less desirable is their very high purchase cost and shorter lifespan.
Many batteries contain toxic chemicals and/or heavy metals, two things that we really don’t want getting into the environment. As the battery market grows year on year, so does the amount of battery waste being produced.
The two main issues are toxic metals reaching the environment and improperly handled used batteries not being recycled correctly.
Toxic metals
Dead batteries are not usable by the consumer, but they still have the ability to cause harm to the environment. Used batteries may contain lead, nickel, cadmium, cobalt, and manganese, all considered to be toxic heavy metals.
If thrown out in municipal trash, these metals can leach from the used batteries and end up in the soil, groundwater, or the air. Ultimately, they will reach the human food chain with potentially negative consequences for human health. For instance, lead exposure in infants leads to brain development issues.
Luckily the majority of lead acid batteries (commonly used in autos) are recycled and are fairly straightforward to recycle, although this is not without its risks and environmental impacts.
Improper recycling
Lithium is also increasingly disposed of as Li-ion batteries are replaced by consumers and, while Lithium is not classified as a toxic metal, the sheer amount of Lithium-ion batteries now heading to landfill when they should really be recycled is a concern. Especially as these ‘zombie batteries’ are flammable and are responsible for numerous fires in waste and recycling facilities around the world each year. Unless carefully handled, used Li-ion batteries can catch fire or explode if crushed, punctured, or contaminated with water, a likely occurrence if they’re thrown into the trash and not recycled with care.
It is not only Lithium-ion batteries which may pose a fire risk. Used batteries of any type, even primary alkaline batteries, may contain residual charge. If their terminals come into contact with other batteries or metals they may short-circuit while in the trash or recycling facility, therefore extra care is required from the consumer not to just toss used batteries in the trash or a bin. Experts suggest covering their terminals with tape or packing them together in such a way that they can’t come into contact with other materials.
Where to recycle used batteries?
Some personal responsibility has to fall on the shoulders of the consumer here as batteries are actually not that difficult to recycle these days in many countries.
Here are some options:
- Many supermarkets and DIY stores provide recycling bins for small used batteries of all types associated with smaller electronic devices.
- Municipal waste sites will take batteries, even larger batteries such as lead-acid car batteries and Li-ion laptop batteries.
- Specialized recycling organizations, such as Call2Recycle in the USA, will take batteries or provide drop-off points in certain retailers.
It only takes a very brief search engine search to find options for your region or country.
You may hear both terms used and they’re connected.
A cell is a single power source that provides a set amount of DC voltage, for example, a watch battery is a single cell that has a low voltage of 1.5 to 3V.
Whereas, a full battery is often a ‘stack’ of cells added together in order to create the higher voltage needed to run larger devices. This can be anything from a laptop battery to one in an electronic vehicle. Also, if you consider a mobile phone’s power bank – this is essentially a bunch of cells packed together in series to give a large storage capacity for power on the go.
The terms LFP, NMC, and NCA all refer to different chemistries commonly used in lithium secondary cells.
- LFP: lithium iron phosphate
- NMC: lithium nickel manganese cobalt
oxide - NCA: lithium nickel cobalt aluminum oxide
Between 2015 to 2018 NMC pouch and prismatic cells, which are commonly found in smartphones and laptops to this day, and, to a lesser extent, cylindrical NCA cells were the dominant forces in the secondary cell market.
But as we head into the 2020s the older LFP technology is making a comeback in larger cells which are now becoming important due to the growing EV market. Tesla, for example, utilizes large LFP cells.
Challenges are that larger prismatic cells may grow hotter and so a cooling system for them is required, for instance, liquid cooling. But they are not as fragile as pouch cells, and, due to their size are, today in 2021, able to hold upwards of 200 Ah capacity each.
Glossary of key battery terminology
Amp(ere) Hour – a unit of measurement of a battery’s electrical storage capacity. Current multiplied by time in hours equals ampere-hours. One amp hour is equal to a current of one ampere flowing through the circuit for one hour. Also, 1 amp hour is equal to 1,000 mAh.
Battery – this is an electrochemical device which stores energy. A battery is usually a series of cells which are connected in order to provide a larger amount of energy cumulatively.
Battery capacity – the total power output of a cell, often measured in mAh for electronic devices.
Cell – this is a single electrochemical device often combined with others to create a battery. They consist of positive and negative plates and a form of electrolyte which can store and release energy.
Charge – when we charge a battery we’re converting an electric current into chemical energy which is stored within it.
Charge rate – this is the current applied to the battery when charging. This is a fraction of the battery’s overall capacity, expressed as C/3, for example.
Cycle – one charge/discharge sequence.
Cycle life – Some secondary cells have a finite number of cycles as their lifespan, this is their cycle life. After they reach this number, their capacity is likely to be very poor.
Discharge – where a battery converts its chemical energy into electrical energy to power a device.
Electrolyte – inside a battery a chemical compound, such as Lithium, will be dissolved into a solvent of some kind (perhaps water) and this electrolyte contains ions which conducts electrical current.
Internal resistance – the resistance to the flow of electrical current inside the cell or battery. This is likely to increase as a battery ages.
Ohm’s law – shows the amount of current flowing through a circuit in Amperes. Current (I) is calculated by dividing the pressure in volts (V) by the resistance in ohms (R), but the calculation can also find the voltage and resistance in the circuit, too.
- Current (I) = V/R
- Voltage (V) = I x R
- Resistance (R) = V/I
Rated capacity – amp-hours the cell is able to provide under specific conditions which are set by the manufacturer. This will be in the battery specification sheet. The conditions will include elements such as rate of discharge, end voltage, and temperature.
Rechargeable – secondary batteries or cells which are able to be charged and discharged repeatedly.
Volt – a measure of electromotive force or difference of potential which causes a current of one amp to flow through a resistance of one ohm.
Watt – a measure of total power which is amps x volts. W = A x V.
So, if the current is 5 amps and the voltage is 110V, the wattage (or total power) is 550W.
Do you need help to get better results from your Chinese suppliers?
Speak with us now. Hit the button below to request a consultation.












